 The first can be formed from an element with two valence electrons in its outer shell, like lithium: It is the unhybridized p orbitals that then form pi bonds for double bonding:Īgain there are two ways to form sp hybrids. Or if the atom has more than three valence electrons in its outer shell three of the electron orbitals hybridize and one of the p orbitals remains unhybridized: 1) hybridization of an element with three valence electrons in its outer shell, like boron will yield three full sp 2 hybrid orbitals and no left over electrons. There are two ways to form sp 2 hybrid orbitals that result in two types of bonding. Other hybridizations follow the same format. When these sp 3 hybrid orbitals overlap with the s orbitals of the hydrogens in methane, you get four identical bonds, which is what we see in nature. We take the two higher energy p orbital electrons and the two lower energy s orbital electrons and meld them into four equal energy sp 3 ( 1s 3 p orbitals = sp 3) hybrid orbitals. So even though the bonds are made up of different energy orbitals they make all the same type of bonds, how can this be? Well, the way we explain it is hybridization. All the bond lengths and strengths in methane are roughly the same. So the structure would look like this:īut we know this is not what methane (CH 4) actually looks like. So in a molecule of CH 4 you should see two long bonds between the s-s orbital overlaps, and two shorter bonds between the p-s orbital overlaps. Now, remembering back to the atomic theory, we know that s orbitals are of lower energy than p orbitals, correct? So that means when they bond to other atoms, the p orbital electrons would form stronger (higher energy bonds) than the s orbital electrons. 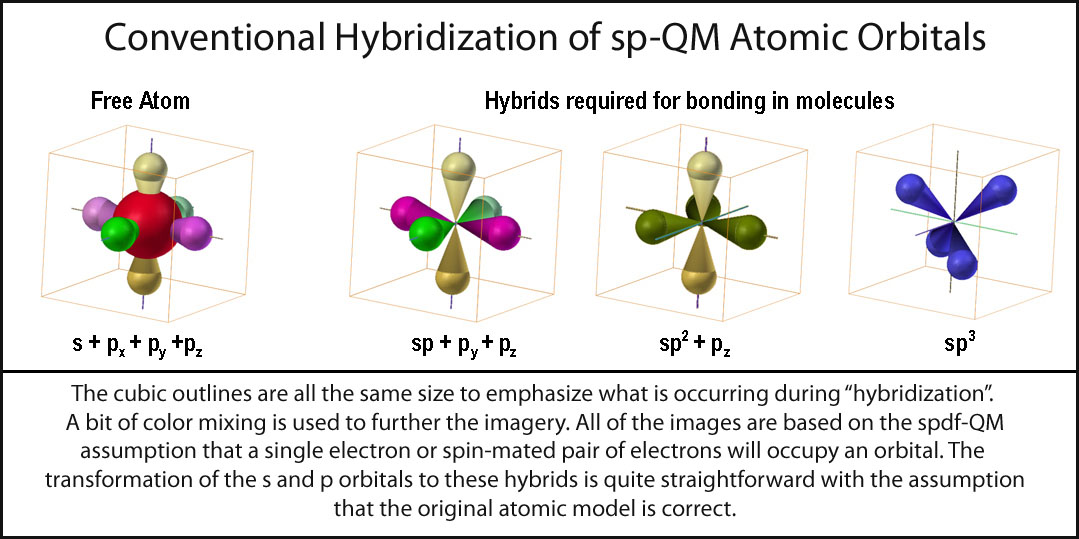
Here is what I mean: Carbon has an electron configuration of 1s 2 2s 2 2p 2 There are four valence electrons in carbon's outermost shell that can bond: two s orbital electrons and 2 p orbital electrons. When we talk about hybrid orbitals we are visualizing what we believe must occur within a molecules bonding structure to result in the molecular structures we can see. .jpg)
Scientists hybridize plants all the time to give them better taste, more resilience to disease etc. What is a hybrid? Well, when you combine two things into one that is a hybrid. Pi bonds are found in double and triple bond structures. Another type of bond, a pi (p) bond is formed when two p orbitals overlap. This overlap may involve s-s, s-p, s-d or even p-d orbitals. Single covalent bonds that form between nuclei are created from the "head-to-head" overlap of orbitals and are called sigma ( s) bonds. This simply means that electron density is highest along the axis of the bond. When the bond forms, the probabiity of finding electrons changes to become higher within the region of space between the two nuclei. The valence orbitals of an atom surrounded by a tetrahedral arrangement of bonding pairs and lone pairs consist of a set of four sp 3 hybrid orbital.Valence bond theory is an empirically derived theory that describes how orbitals overlap in molecules to form bonds. A lone pair, an unpaired electron, a single bond, or a multiple bond would each count as one region of electron density. As we know from the discussion of VSEPR theory, a region of electron density contains all of the electrons that point in one direction. The central atom(s) in each of the structures shown contain three regions of electron density and are sp2 hybridized. 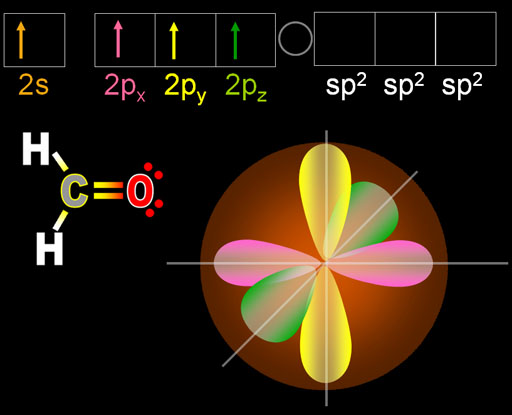
Valence bond theory would predict that the two \ce.įigure 7.5.9. Oxygen has the electron configuration 1 s 22 s 22 p 4, with two unpaired electrons (one in each of the two 2p orbitals). As an example, let us consider the water molecule, in which we have one oxygen atom bonding to two hydrogen atoms. However, to understand how molecules with more than two atoms form stable bonds, we require a more detailed model. Thinking in terms of overlapping atomic orbitals is one way for us to explain how chemical bonds form in diatomic molecules. This is not consistent with experimental evidence. The hypothetical overlap of two of the 2p orbitals on an oxygen atom (red) with the 1s orbitals of two hydrogen atoms (blue) would produce a bond angle of 90°.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
